Blood Conservation and Management in Cardiac Surgery
Now it's more important than ever and you can directly affect it easily!
There really are only 2 questions:
-
Are you and your team really serious about saving blood & avoiding transfusions? And if so...
-
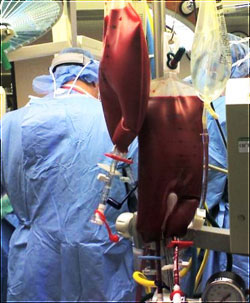
Can you limit the amount of blood loss & hemodilution and reverse it?
The Hemobag® does both.
Eight (8) articles (whose abstracts are provided below) really sum up the factors effecting blood use and cardiac surgery today. From the shortage to the wastage to the adverse effects, this news cannot be more timely or more to the point. We as professionals all know that extreme hemodilution is a bad thing not only for oxygen delivery (dilutional anemia) but also for hemostasis (dilutional coagulopathy) and homeostasis (edema and organ dysfunction). There is increasing evidence that albumin levels possess additional effects on organ perfusion, microcirculation, tissue oxygenation, inflammation, endothelial activation, capillary leakage, and tissue edema that are well beyond just the volume expanding/replacing properties.
This study, which was stopped at Duke, shows that extreme hemodilution is detrimental, but I dare say that we all tend to have patients with native on CPB Hgb levels that are between 7-9 g/dL most of the time. Currently there are studies to narrow safe triggers in coronary disease.
The silver bullet native Hct's for all comers from the literature for no adverse effects appears to be 22% on Pump and 24% out of the room into the ICU. Below these points the literature has shown increased morbidity and mortality. Transfusing allogeneic RBCs in general, but above these optimal native Hct levels, has also shown to too high immediately post-op is also not good as well.
In your reading time, we have put these articles together to help try and paint what's going on in the world, and to try and keep us all together on the same page at the same time with regards to obtaining the best patient outcomes through solid blood management and optimal autologous whole blood protection and use while avoiding misuse and wastage.
So below is a PPT presentation on a "Multidisciplinary Multimodality approach to Blood Conservation in Cardiac Surgery", and an Abstract Argument titled, "Blood, Ethics and Common Sense Best Practices in Cardiac Surgery" for all concerned about blood to contemplate the shifts and changes in sizes and triggers and plain old common sense saving of native autologous whole blood. Don't forget to talk to your surgeons and anesthesiologists about the Hemobag® to help improve postoperative coagulation and enhance patient stability and recovery from fluid shifts at the microcirculatory level.
Enjoy the reading, and feel free to send professional comments or questions to info@mybloodfirst.com
-Global Blood Resources (GBR)
The association of perioperative red blood cell transfusions and decreased long-term survival after cardiac surgery
BACKGROUND:
Exposure to red blood cell (RBC) transfusions has been associated with increased mortality after cardiac surgery. We examined long-term survival for cardiac surgical patients who received one or two RBC units during index hospitalization.
METHODS:
Nine thousand seventy-nine consecutive patients undergoing coronary artery bypass graft, valve, or coronary artery bypass graft/valve surgery at eight centers in northern New England during 2001-2004 were examined after exclusions. A probabilistic match between the regional registry and the Social Security Administration's Death Master File determined mortality through June 30, 2006. Cox Proportional Hazard and propensity methods were used to calculate adjusted hazard ratios.
RESULTS:
Thirty-six percent of patients (n = 3254) were exposed to one or two RBC units. Forty-three percent of RBCs were given intraoperatively, 56% in the postoperative period and 1% were preoperative. Patients transfused were more likely to be anemic, older, smaller, female and with more comorbid illness. Survival was significantly decreased for all patients exposed to 1 or 2 U of RBCs during hospitalization for cardiac surgery compared with those who received none (P < 0.001). After adjustment for patient and disease characteristics, patients exposed to 1 or 2 U of RBCs had a 16% higher long-term mortality risk (adjusted hazard ratios = 1.16, 95% CI: 1.01-1.34, P = 0.035).
CONCLUSIONS:
Exposure to 1 or 2 U of RBCs was associated with a 16% increased hazard of decreased survival after cardiac surgery.
Intraoperative transfusion of 1 U to 2 U packed red blood cells is associated with increased 30-day mortality, surgical-site infection, pneumonia, and sepsis in general surgery patients
BACKGROUND:
Transfusion of packed red blood cells (PRBCs) increases morbidity and mortality in select surgical specialty patients. The impact of low-volume, leukoreduced RBC transfusion on general surgery patients is less well understood.
STUDY DESIGN:
The American College of Surgeons National Surgical Quality Improvement Program participant use file was queried for general surgery patients recorded in 2005 to 2006 (n = 125,223). Thirty-day morbidity (21 uniformly defined complications) and mortality, demographic, preoperative, and intraoperative risk variables were obtained. Infectious complications and composite morbidity and mortality were stratified across intraoperative PRBCs units received. Multivariable logistic regression was used to assess influence of transfusion on outcomes, while adjusting for transfusion propensity, procedure type, wound class, operative duration, and 30+ patient risk factors.
RESULTS:
| 1 U | 2 U | |
| 30-day Mortality | 1.32 | 1.38 |
| Composite Morbidity | 1.23 | 1.40 |
| Pneumonia | 1.24 | 1.25 |
| Sepsis/Shock | 1.29 | 1.53 |
After adjustment for transfusion propensity, procedure group, wound class, operative duration, and all other important risk variables, 1 U PRBCs significantly (p < 0.05) increased risk of 30-day mortality (odds ratio [OR] = 1.32), composite morbidity (OR = 1.23), pneumonia (OR = 1.24), and sepsis/shock (OR = 1.29). Transfusion of 2 U additionally increased risk for these outcomes (OR = 1.38, 1.40, 1.25, 1.53, respectively; p <or= 0.05) plus surgical-site infection (OR = 1.25; p < 0.05). A risk index for calculating transfusion likelihood demonstrated very good discrimination (c-index = 0.844).
CONCLUSIONS:
Intraoperative transfusion of PRBCs increases risk for mortality and several morbidities in general surgery patients. These risks, substantial for even 1 U, remain after adjustment for transfusion propensity and numerous risk factors available in the American College of Surgeons National Surgical Quality Improvement Program. Transfusion for mildly hypovolemic or anemic patients should be discouraged in light of these risks.
Duration of red-cell storage and complications after cardiac surgery
BACKGROUND:
Stored red cells undergo progressive structural and functional changes over time. We tested the hypothesis that serious complications and mortality after cardiac surgery are increased when transfused red cells are stored for more than 2 weeks.
METHODS:
We examined data from patients given red-cell transfusions during coronary-artery bypass grafting, heart-valve surgery, or both between June 30, 1998, and January 30, 2006. A total of 2872 patients received 8802 units of blood that had been stored for 14 days or less ("newer blood"), and 3130 patients received 10,782 units of blood that had been stored for more than 14 days ("older blood"). Multivariable logistic regression with propensity-score methods was used to examine the effect of the duration of storage on outcomes. Survival was estimated by the Kaplan-Meier method and Blackstone's decomposition method.
RESULTS:
| 20 days | 11 days | |
| In-Hostpital Mortality | 2.8% | 1.7% |
| Intubation 72+ Hours | 9.7% | 5.6% |
| Renal Failure | 2.7% | 1.6% |
| Sepsis or Septicemia | 4.0% | 2.8% |
The median duration of storage was 11 days for newer blood and 20 days for older blood. Patients who were given older units had higher rates of in-hospital mortality (2.8% vs. 1.7%, P=0.004), intubation beyond 72 hours (9.7% vs. 5.6%, P<0.001), renal failure (2.7% vs. 1.6%, P=0.003), and sepsis or septicemia (4.0% vs. 2.8%, P=0.01). A composite of complications was more common in patients given older blood (25.9% vs. 22.4%, P=0.001). Similarly, older blood was associated with an increase in the risk-adjusted rate of the composite outcome (P=0.03). At 1 year, mortality was significantly less in patients given newer blood (7.4% vs. 11.0%, P<0.001).
CONCLUSIONS:
In patients undergoing cardiac surgery, transfusion of red cells that had been stored for more than 2 weeks was associated with a significantly increased risk of postoperative complications as well as reduced short-term and long-term survival.
Increased mortality, postoperative morbidity, and cost after red blood cell transfusion in patients having cardiac surgery
BACKGROUND:
Red blood cell transfusion can both benefit and harm. To inform decisions about transfusion, we aimed to quantify associations of transfusion with clinical outcomes and cost in patients having cardiac surgery.
METHODS:
Clinical, hematology, and blood transfusion databases were linked with the UK population register. Additional hematocrit information was obtained from intensive care unit charts. Composite infection (respiratory or wound infection or septicemia) and ischemic outcomes (myocardial infarction, stroke, renal impairment, or failure) were pre-specified as co-primary end points. Secondary outcomes were resource use, cost, and survival. Associations were estimated by regression modeling with adjustment for potential confounding. All adult patients having cardiac surgery between April 1, 1996, and December 31, 2003, with key exposure and outcome data were included (98%).
RESULTS:
Adjusted odds ratios for composite infection (737 of 8516) and ischemic outcomes (832 of 8518) for transfused versus nontransfused patients were 3.38 (95% confidence interval [CI], 2.60 to 4.40) and 3.35 (95% CI, 2.68 to 4.35), respectively. Transfusion was associated with increased relative cost of admission (any transfusion, 1.42 times [95% CI, 1.37 to 1.46], varying from 1.11 for 1 U to 3.35 for >9 U). At any time after their operations, transfused patients were less likely to have been discharged from hospital (hazard ratio [HR], 0.63; 95% CI, 0.60 to 0.67) and were more likely to have died (0 to 30 days: HR, 6.69; 95% CI, 3.66 to 15.1; 31 days to 1 year: HR, 2.59; 95% CI, 1.68 to 4.17; >1 year: HR, 1.32; 95% CI, 1.08 to 1.64).
CONCLUSIONS:
Red blood cell transfusion in patients having cardiac surgery is strongly associated with both infection and ischemic postoperative morbidity, hospital stay, increased early and late mortality, and hospital costs.
Influence of intraoperative fluid volume on cardiopulmonary bypass hematocrit and blood transfusions in coronary artery bypass surgery
BACKGROUND:
A hematocrit (Hct) of less than 25% during cardiopulmonary bypass (CPB) and transfusion of homologous packed red blood cells (PRBC) are each associated with an increased probability of adverse events in cardiac surgery. Although the CPB circuit is a major contributor to hemodilution intravenous (IV) fluid volume may also significantly influence the level of hemodilution. The objective of this study was to explore the influence of asanguinous IV fluid volume on CPB Hct and intraoperative PRBC transfusion.
RESULTS:
After Institutional Review Board approval, a retrospective chart review of 90 adult patients that had undergone an elective, isolated CABG with CPB was conducted. Regression analysis was used to determine if pre-CPB fluid volume was associated with the lowest CPB Hct and the incidence of an intraoperative PRBC transfusion. In separate multivariate analyses, higher pre-CPB fluid volume was associated with lower minimum CPB Hct (p < .0001), and higher minimum CPB Hct was associated with a decreased probability of PRBC transfusion (p < .0001). Compared to patients that received <1600 mL (n = 55) of pre-CPB fluid, those that received >1600 mL (n = 35) had a decreased mean low CPB Hct (22.4% vs 25.6%, p < .0001), an increased incidence of a CPB Hct <25% (74% vs. 38%, p = .0008) and PRBC transfusion (60% vs. 16%, p < .0001), and increased median PRBC units transfused (2.0 vs 1.0, p = .1446) despite no significant difference in gender, age, patient size, baseline Hct, or CPB prime volume. Patients that received a PRBC transfusion (n = 30) received a significantly higher volume of pre-CPB fluid than nontransfused patients (1800 vs. 1350 mL, p = .0039).
CONCLUSION:
These findings suggest that pre-CPB fluid volume can significantly contribute to hemodilutional anemia in cardiac surgery. Optimizing pre-CPB volume may preserve baseline Hct and help limit intraoperative hemodilution.
The Cardiotomy Trial: A randomized, double-blind study to assess the effect of processing of shed blood during cardiopulmonary bypass on transfusion and neurocognitive function
BACKGROUND:
Reinfusion of unprocessed cardiotomy blood during cardiac surgery can introduce particulate material into the cardiopulmonary bypass circuit, which may contribute to postoperative cognitive dysfunction. On the other hand, processing of this blood by centrifugation and filtration removes coagulation factors and may potentially contribute to coagulopathy. We sought to evaluate the effects of cardiotomy blood processing on blood product use and neurocognitive functioning after cardiac surgery.
METHODS AND RESULTS:
Patients undergoing coronary and/or aortic valve surgery using cardiopulmonary bypass were randomized to receive unprocessed blood (control, n=134) or cardiotomy blood that had been processed by centrifugal washing and lipid filtration (treatment, n=132). Patients and treating physicians were blinded to treatment assignment. A strict transfusion protocol was followed. Blood transfusion data were analyzed using Poisson regression models. The treatment group received more intraoperative red blood cell transfusions (0.23+/-0.69 U versus 0.08+/-0.34 U, P=0.004). Both red blood cell and non-red blood cell blood product use was greater in the treatment group and postoperative bleeding was greater in the treatment group. Patients were monitored intraoperatively by transcranial Doppler and they underwent neuropsychometric testing before surgery and at 5 days and 3 months after surgery. There was no difference in the incidence of postoperative cognitive dysfunction in the 2 groups (relative risk: 1.16, 95% CI: 0.86 to 1.57 at 5 days postoperatively; relative risk: 1.05, 95% CI: 0.58 to 1.90 at 3 months). There was no difference in the quality of life nor was there a difference in the number of emboli detected in the 2 groups.
CONCLUSIONS:
Contrary to expectations, processing of cardiotomy blood before reinfusion results in greater blood product use with greater postoperative bleeding in patients undergoing cardiac surgery. There is no clinical evidence of any neurologic benefit with this approach in terms of postoperative cognitive function.
Changing age distribution of the blood donor population in the United States
BACKGROUND:
The American Red Cross has been maintaining a research database of all blood donors. Such a database provides a unique opportunity for monitoring changes over time in donor and donation patterns.
STUDY DESIGN AND METHODS:
Changes in age distribution among blood donors were analyzed through comparison of the volunteer donor population in 1996, 1999, 2002, and 2005, before and after adjustment for demographic changes of the general population in the United States.
RESULTS:
Donations by repeat donors 50 years or older as a proportion of total donations increased from 22.1 percent in 1996 to 34.5 percent in 2005, or 1.4 percent per year, whereas donations from repeat donors of 25 to 49 years decreased from 49.1 percent in 1996 to 37.1 percent in 2005, or 1.3 percent per year. After adjusting for general population trends, the effective number of donors decreased by more than 10 percent in female and male repeat donors of age 20 to 49 years and male first-time donors of age 25 to 49 years from 1996 to 2005; female and male repeat donors of age 25 to 39 years decreased by greater than 40 percent. Prevalence rates of major infectious disease markers decreased by 3.3 percent or more per year for first-time donations and by 6.4 percent or more per year for repeat donations.
CONCLUSION:
The aging patterns of blood donors suggest the need for improved recruitment and retention in the young adult and middle-aged groups. A severe shortage of blood and blood components may be forecast in the foreseeable future unless offset by significant increased supply or reduced usage of blood and blood components.
Ultrafiltration reduces blood transfusions following cardiac surgery: A meta-analysis
BACKGROUND:
Although used routinely in pediatric patients, ultrafiltration techniques that reverse hemodilution are infrequently used in adults. Data from small, unblinded clinical trials suggest that the use of ultrafiltration can reduce inflammatory mediators, improve cardiac function, and reduce hemodilution. We conducted a meta-analysis of randomized trials to evaluate the effects of ultrafiltration on blood transfusions and blood loss following adult cardiac surgery.
METHODS:
Medline, EMBASE, and Cochrane databases were searched and randomized controlled trials evaluating modified and/or conventional ultrafiltration, meeting pre-determined selection criteria, were obtained. Quality evaluation and data extraction were performed by two independent observers blinded to study source. Random effects models were used to determine pooled effect estimates and sources of heterogeneity were explored using meta-regression.
RESULTS:
One hundred and thirty two studies were screened and 10 randomized trials evaluating 1004 patients (control, n = 495; ultrafiltration, n = 509) were identified of which only two were double-blinded. The use of ultrafiltration was associated with a reduction in postoperative blood transfusions (weighted mean difference [95% CI] of -0.73 units [-1.16, -0.31]; p = 0.001). This reduction was greater in studies evaluating modified ultrafiltration. Use of ultrafiltration was also associated with reduced postoperative bleeding (-70 ml, [-118, -21]; p = 0.005), which was driven primarily by trials evaluating modified rather than conventional ultrafiltration.
CONCLUSIONS:
Use of ultrafiltration is associated with a significant reduction in postoperative blood transfusions as well as reduced bleeding in adults undergoing cardiac surgery. The efficacy and cost-effectiveness of ultrafiltration as a blood conservations strategy should be evaluated in a large, randomized, double-blinded study.
Can you limit the amount of blood loss
and hemodilution and reverse it?
The Hemobag® does both.